Test Principle
The test is divided in two steps:
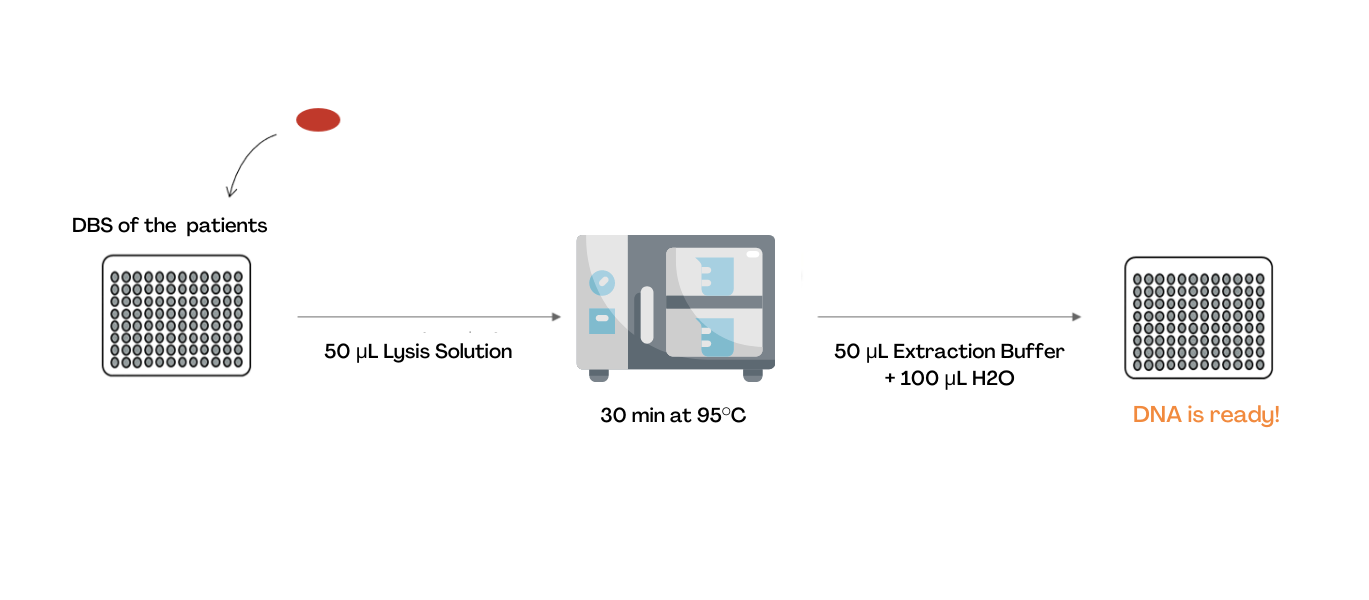
- Extraction and lysis of the sample
DNA is obtained from dried blood spot (DBS) via the consecutive addition of two buffers and an incubation step at 95°C.
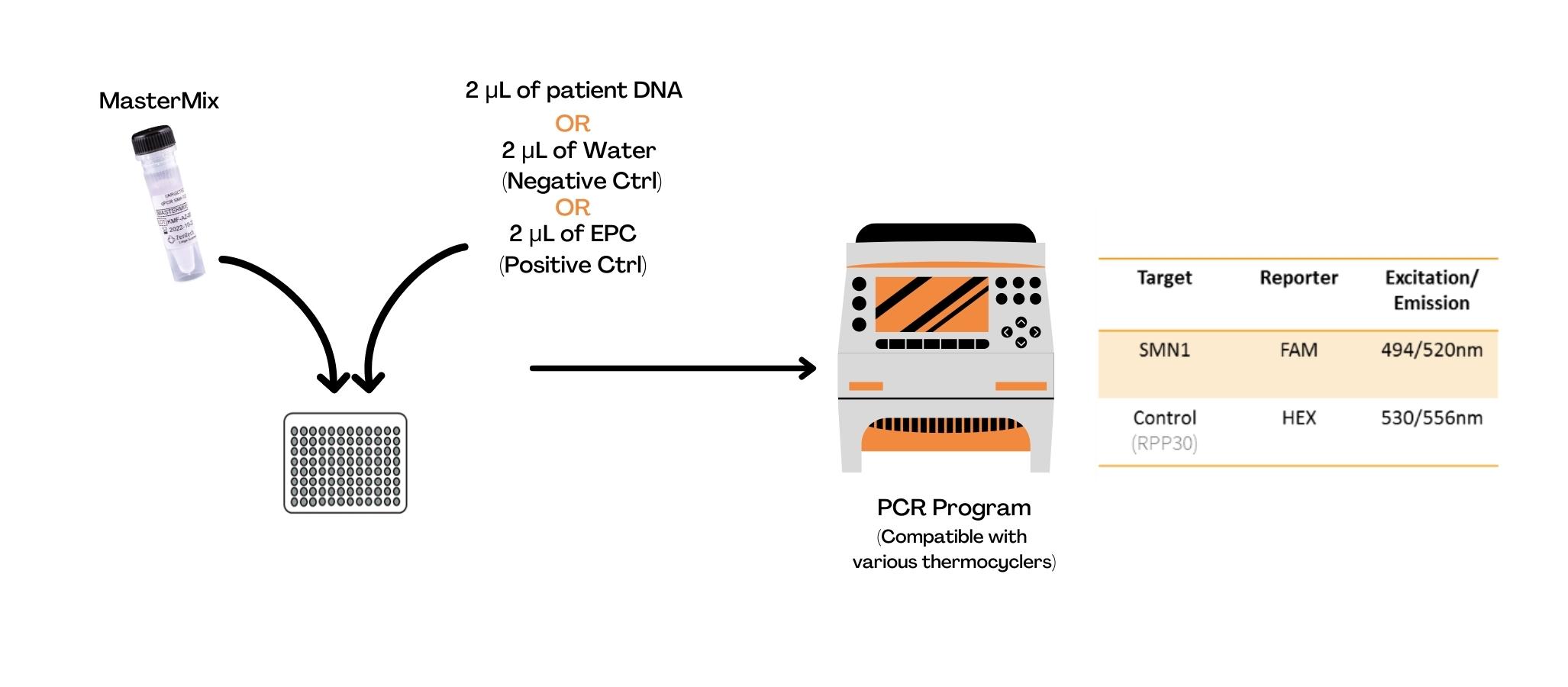
- DNA amplification by qPCR
The extracted sample and the ready-to-use Master Mix are transferred to a plate compatible with the thermocycler. The qPCR amplification step takes approximately 1 hour and can be performed on multiple thermocyclers (Bio-Rad CFX96, ThermoFisher QuantStudio™ 5, Roche LightCycler® 480).
The non-mutated SMN1 allele and the RPP30 reference gene are detected by the FAM and HEX fluorochromes, respectively.
The results can be interpreted as follows:
|
|
FAM
|
HEX
|
| Healthy or heterozygous patient |
+
|
+
|
|
SMA-affected patient
|
-
|
+
|
The correct execution of the amplification step is guaranteed by an external positive control made of synthetic DNA which is provided in the kit and recognized by HEX and FAM.
Illness
This test is designed to genotype the SMN1 mutation site for newborn screening of Anterior Spinal Muscular Atrophy (SMA) from a blood sample dried on 903® or 226 blotting paper.
General Info
Product code: K-MF-480 for 480 determinations